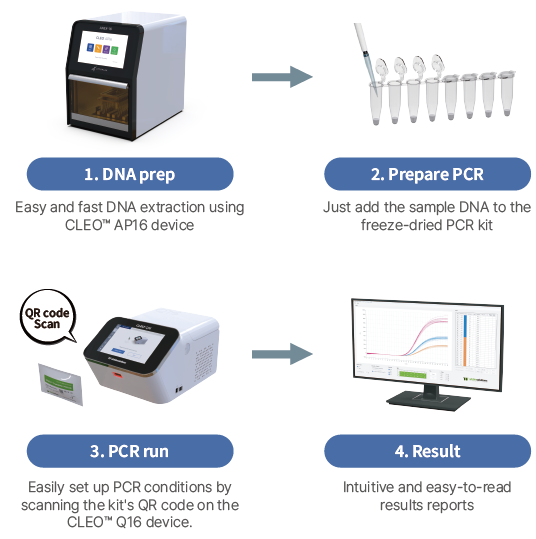
| Principle of Procedure |
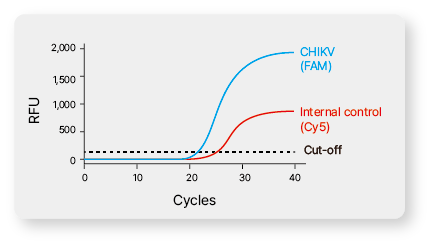
Real-time PCR |
| Target |
FAM for the sample, CY5 for ther internal control |
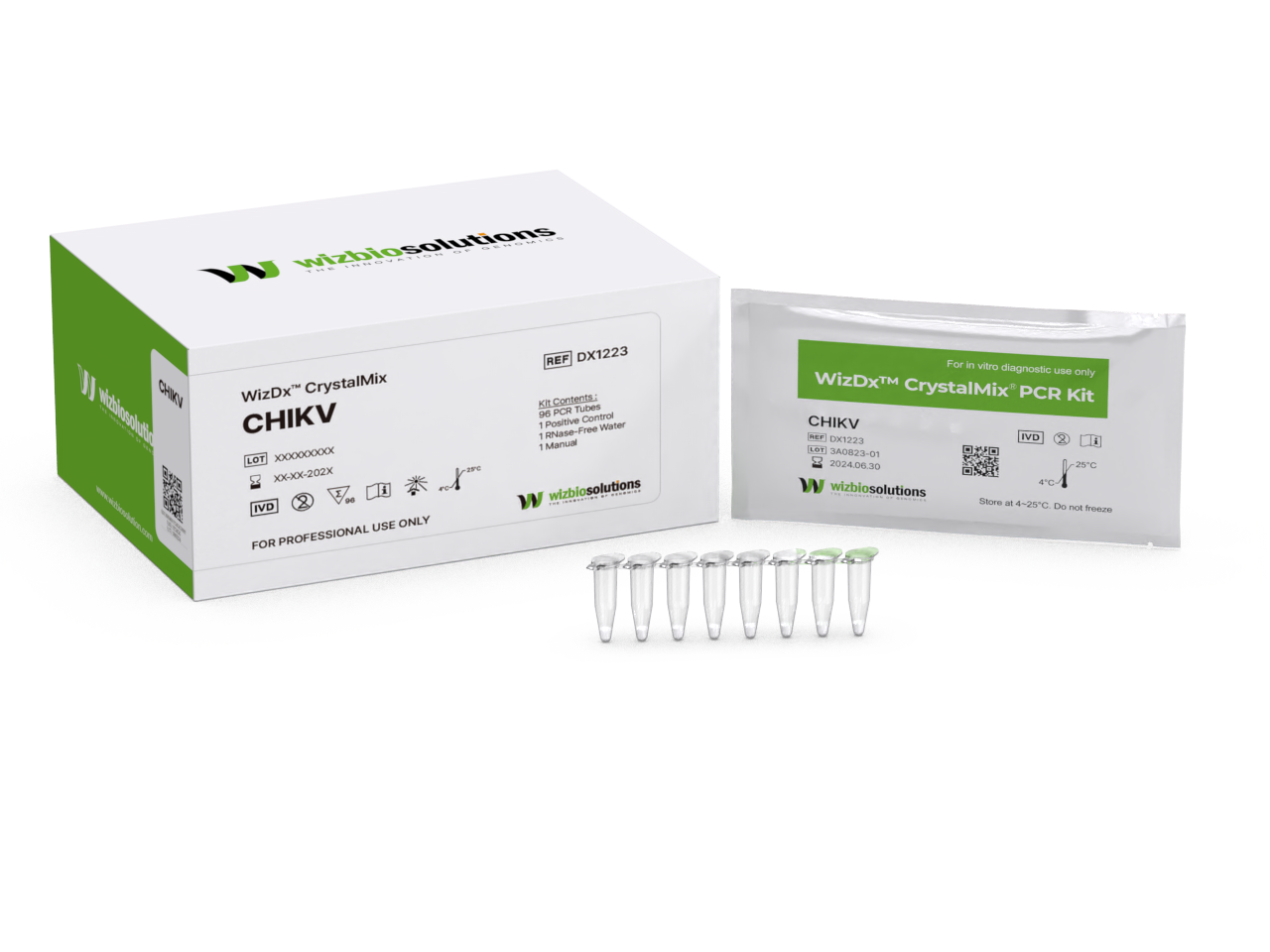
| Tests for Package |
96 Tests/kit |
| Reagent Type |
Lyophilized (Freeze-dried) in PCR tube |
| Limit of Detection (LoD) |
0.25 copy/μL |
| Precision |
Respectively detect the reference specimen of high and low concentrations in
different time ranges-10 times, and the precision values of intra-assay and
inter-assay Ct values were all <5%. |
| Specificity |
No cross-reactivity of the assay was observed with Dengue virus, Plasmodium
vivax, Japanese Encephalitis virus, SARS-CoV-2, Human Adenovirus Type 5,
Respiratory syncytial virus A, Influenza A (H3N2), Zika virus (African), Measles virus. |
| Sample Types |
Serum and plasma (EDTA) |
| Shipping/Storage |
Shipping and Storage at room temperature for 12 months |
| Compatible Instruments |
CLEO™ Q16 (Wizbiosolutions), CFX96(Bio-Rad) |