| Principle of Procedure |
Real-time PCR. |
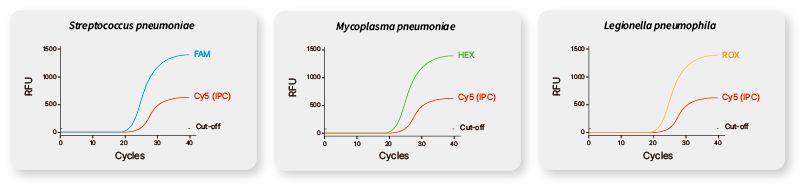
| Target |
FAM for Streptococcus pneumoniae, HEX for Mycoplasma pneumoniae,
ROX for Legionella pneumophila, Cy5 for Internal Control. |
| Tests for Package |
96 Tests/kit. |
| Reagent Type |
Lyophilized (Freeze-dried) in PCR tube. |
| Limit of Detection (LoD) |
0.75 copies/μL. |
| Precision |
Respectively detect the reference specimen of high and low concentrations in
different time ranges-10 times, and the precision values of intra-assay and inter-assay
Ct values were all < 5%. |
| Specificity |
For analytical specificity, cross-reactivity was evaluated by testing related pathogens
and microorganisms that are likely to be present in the cervical, vaginal, and genital.
The test was repeated three times and showed no cross-reactivity. |
| Sample Types |
Oropharyngeal and nasopharyngeal swabs or sputum specimens. |
| Shipping/Storage |
Shipping and Storage at room temperature for 12 months. |
| Compatible Instruments |
CLEO™ Q16 (Wizbiosolutions), CFX96 (Bio-Rad), QuantStudio™ 5 (ThermoFisher). |